|
|
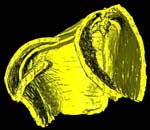
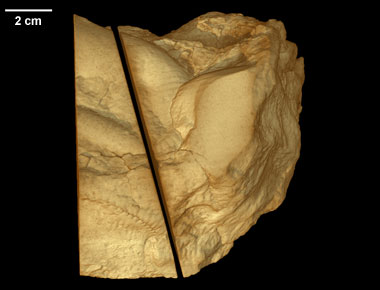
Pteridinium simplex , Ediacaran Fossil , Ediacaran Fossil
Dr. Michael Meyer
-
Christopher Newport University
D. Elliott, A.D. Wood, N.F. Polys, M. Colbert, J.A. Maisano, P. Vickers-Rich, M. Hall, K.H. Hoffman, G. Schneider, and S. Xiao
|
|

|
 |
|
|
|
|
|

|
Image processing: Dr. Michael Meyer
Image processing: Dr. Jessie Maisano
Publication Date:
04 Jun 2014
|
|
|
This page serves supplemental imagery for a paper entitled Three-dimensional microCT analysis of the Ediacara fossil Pteridinium simplex sheds new light on its ecology and phylogenetic affinity, by M. Meyer, D. Elliott, A.D. Wood, N.F. Polys, M. Colbert, J.A. Maisano, P. Vickers-Rich, M. Hall, K.H. Hoffman, G. Schneider, and S. Xiao (Precambrian Research, 249, 79-87). The abstract is as follows:
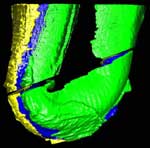
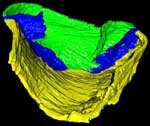
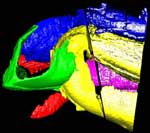
Ediacara fossils often exhibit enigmatic taphonomy that complicates morphological characterization and ecological and phylogenetic interpretation; such is the case with Pteridinium simplex from the late Ediacaran Kliphoek Member in southern Namibia. Pteridinium simplex is often preserved as three-dimensional (3D) casts and molds in coarse-grained quartzites, making detailed morphological characterization difficult. In addition, P. simplex is often transported, distorted, and embedded in gutter fills or channel deposits, further obscuring its morphologies. By utilizing microfocus X-ray computed tomography (microCT) techniques, we are able to trace individual specimens and their vanes in order to digitally restore the 3D morphology of this enigmatic fossil. Our analysis shows that P. simplex has a very flexible integument that can be bent, folded, twisted, stretched, and torn, indicating a certain degree of elasticity. We find no evidence for vane identity change or penetrative growth that were previously used as evidence to support a fully endobenthic lifestyle of P. simplex; instead, the traditional interpretation of a semi-endobenthic or epibenthic lifestyle is favored. The elastic integument of P. simplex is inconsistent with a phylogenetic affinity with xenophyophore protists; instead, its physical property is consistent with the presence of collagen and cellulose, an inference that would provide constraints on the phylogenetic affinity of P. simplex.
Click here to download the original scan data for slab 2 (1.8 Gb) and here to download the original scan data for slab 3 (1.5 Gb).

About the Species
This specimen was collected from the upper Kliphoek Member of the Dabis Formation, Kuibis Subgroup, located at Aar Farm in the Aus region of southern Namibia. It was made available to the University of Texas High-Resolution X-ray CT Facility for scanning by Dr. Michael Meyer of Western Carolina University and Dr. Shuhai Xiao of Virginia Tech. Funding for scanning was provided by Dr. Xiao (NSF grant EAR-0844235).

About this Specimen
This specimen was scanned by Matthew Colbert as two separate slabs. Slab 2 (smaller piece) was scanned on 12 June 2012 horizontally for a total of 1312 slices. Each 1024 x 1024 pixel slice is 0.096 mm thick, with an interslice spacing of 0.096 mm and a field of reconstruction of 91 mm. Slab 3 (larger piece) was scanned on 19 September 2012 horizontally for a total of 1083 slices. Each 1024 x 1024 pixel slice is 0.115 mm thick, with an interslice spacing of 0.115 mm and a field of reconstruction of 107 mm.

About the
Scan
Literature
Narbonne GM (2005) The Ediacara Biota: Neoproterozoic origin of animals and their ecosystems. Annu Rev Earth Planet Sci 33:421-442.
Xiao S, Laflamme M (2009) On the eve of animal radiation: Phylogeny, ecology and evolution of the Ediacara biota. Trends Ecol Evol 24(1):31-40.
Schiffbauer JD, Laflamme M (2012) Lagerstätten through time: A collection of exceptional preservational pathways from the terminal Neoproterozoic through today. Palaios 27(5):275-278.
Laflamme M, Schiffbauer JD, Narbonne GM, Briggs DG (2011) Microbial biofilms and the preservation of the Ediacara biota. Lethaia 44(2):203-213.
Cai Y, Schiffbauer JD, Hua H, Xiao S (2012) Preservational modes in the Ediacaran Gaojiashan Lagerstätte: Pyritization, aluminosilicification, and carbonaceous compression. Palaeogeogr Palaeoclimatol Palaeoecol 326-328:109-117.
Gehling JG (1999) Microbial mats in terminal Proterozoic siliciclastics: Ediacaran death masks. Palaios 14(1):40-57.
Seilacher A, Grazhdankin D, Legouta A (2003) Ediacaran biota: The dawn of animal life in the shadow of giant protists. Paleontol Res 7(1):43-54.
Laflamme M, Darroch SAF, Tweedt SM, Peterson KJ, Erwin D (2013) The end of the Ediacara biota: Extinction, biotic replacement, or Cheshire Cat? Gondwana Res 23(2):558-573.
Gürich G (1930) Die bislang ältesten Spuren von Organismen in Südafrika. International Geological Congress South Africa, 1929 (XV) 2:670-680.
Fedonkin MA, Gehling JG, Grey K, Narbonne GM, Vickers-Rich P (2007) The Rise of Animals: Evolution and Diversification of the Kingdom Animalia (Johns Hopkins University Press, Baltimore).
Jenkins RJF (1992) in Origin and Early Evolution of Metazoa, eds Lipps JH & Signor PW (Plenum Press, New York), pp 131-176.
Grazhdankin D, Seilacher A (2002) Underground Vendobionta from Namibia. Palaeontology 45(1):57-78.
Elliott DA, Vickers-Rich P, Trusler P, Hall M (2011) New evidence on the taphonomic context of the Ediacaran Pteridinium. Acta Palaeontol Pol 56(3):641-650.
Glaessner MF, Daily B (1959) The geology and Late Precambrian fauna of the Ediacaran fossil reserve. Record of South Australia Museum 13:369-401.
Pflug HD (1994) Role of size increase in Precambrian organismic evolution. Neues Jahrb Geol Palaontol Abh 193(2):245-286.
Retallack GJ (1994) Were the Ediacaran fossils lichens? Paleobiology 20(4):523-544.
Glaessner MF (1984) The Dawn of Animal Life: A Biohistorical Study (Cambridge Univ. Press, Cambridge, UK).
Vickers-Rich P, et al. (2013) Reconstructing Rangea: New discoveries from the Ediacaran of southern Namibia. J Paleontol 87(1):1-15.
Hall M, et al. (submitted) Changing environments and their impact on metazoan evolution and preservation during the terminal Proterozoic in African Gondwana. Precambrian Res xxx:xxx.
Grotzinger JP, Bowring SA, Saylor BZ, Kaufman AJ (1995) Biostratigraphic and geochronologic constraints on early animal evolution. Science 270(5236):598-604.
Condon D, et al. (2005) U-Pb ages from the Neoproterozoic Doushantuo Formation, China. Science 308(5718):95-98.
Schmitz MD (2012) in The Geologic Time Scale 2012, eds Gradstein FM, Ogg JG, Schmitz MD, & Ogg G (Elsevier, Boston), pp 1045-1082.
Narbonne GM, Xiao S, Shields GA (2012) in Geological Time Scale 2012, eds Gradstein FM, Ogg JG, Schmitz MD, & Ogg G (Elsevier, Oxford), pp 413-435.
Saylor BZ, Kaufman AJ, Grotzinger JP, Urban F (1995) Sequence stratigraphy and sedimentology of the Neoproterozoic Kuibis and Schwarzrand Subgroups (Nama Group), southwestern Namibia. Precambrian Res 73:153-171.
Gresse PG, Germs GJB (1993) The Nama foreland basin: Sedimentation, major unconformity bounded sequences and multisided active margin advance. Precambrian Res 63:247-272.
Meyer M, et al. (submitted) Taphonomy of the Ediacaran fossil Pteridinium simplex preserved three-dimensionally in mass flow deposits in the Nama group of Namibia. J Paleontol xxx:xxx-xxx.
Crimes TP, Fedonkin MA (1996) Biotic changes in platform communities across the Precambrian-Phanerozoic boundary. Rivista Italiana di Paleontologia e Stratigrafia 102(3):317-332.
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671-675.
UNU (2013) Teem: Tools to process and visualize scientific data and images. in UNU.
CIBC (2013) Seg3D: Volumetric image segmentation and visualization. in Seg3D, ed Institute UoUSCaI (University of Utah Scientific Computing and Imaging Institute).
Yoo TS, et al. eds (2002) Engineering and Algorithm Design for an Image Processing API: A Technical Report on ITK - The Insight Toolkit (IOS Press, Amsterdam), pp 586-592.
Sensegraphics (2013) Open source haptics - H3D.org. in H3D, ed Sensegraphics.
Brutzman D, Daly L (2007) X3D: Extensible 3D Graphics for Web Authors (Morgan Kaufmann Publishers Inc., San Francisco, CA).
Consortium WD (2012) Extensible 3D (X3D) specifications: ISO/IEC 19775-1 (Abstract Spec), ISO/IEC 19775-2 (SAI), ISO/IEC 19776-1 (XML Encoding), ISO/IEC 19776-2 (utf-8 Encoding, ISO/IEC), 19776-3 (Binary Encoding). in X3D Spec, ed Consortium WD.
Narbonne GM, Saylor BZ, Grotzinger JP (1997) The youngest Ediacaran fossils from southern Africa. J Paleontol 71(6):953-967.
Dzik J (2003) Anatomical information content in the Ediacaran fossils and their possible zoological affinities. Integr Comp Biol 43(1):114-126.
Dzik J (1999) Organic membranous skeleton of the Precambrian metazoans from Namibia. Geology 27(6):519-522.
Buss LW, Seilacher A (1994) The Phylum Vendobionta: A sister group of the Eumetazoa? Paleobiology 20(1):1-4.
Seilacher A (1992) Vendobionta and Psammocorallia: Lost constructions of Precambrian evolution. J Geol Soc (Lond) 149:607-613.
Retallack GJ (2007) Growth, decay and burial compaction of Dickinsonia, an iconic Ediacaran fossil. Alcheringa 31(3):215-240.
Vogel S (2003) Comparative Biomechanics: Life's Physical World (Princetion University Press, New Jersey, USA).
Hernandez-Nicaise ML, Amsellem J (1982) Ultrastructure of the giant smooth muscle fiber of the ctenophore Beroe ovata. J Ultrastruct Res 72(2):151-168.
Tucker RP, Shibata B, Blankenship TN (2011) Ultrastructure of the mesoglea of the sea anemone Nematostella vectensis (Edwardsiidae). Invertebr Biol 130(1):11-24.
Alexander RM (1971) Connective tissue mechanics of Metridium senile. J Exp Biol 55:775-795.
Alexander RM (1964) Visco-elastic properties of the mesogloea of jellyfish. J Exp Biol 41:363-369.
Zhu M, Gehling JG, Xiao S, Zhao Y-L, Droser M (2008) Eight-armed Ediacara fossil preserved in contrasting taphonomic windows from China and Australia. Geology 36(11):867-870.
Tang F, Bengtson S, Wang Y, Wang X, Yin C (2011) Eoandromeda and the origin of Ctenophora. Evol Dev 13(5):408-414.
Koehl MAR (1999) Ecological biomechanics of benthic organisms: Life history, mechanical design and temporal patterns of mechanical stress. J Exp Biol 202:3469-3476.
Martone PT, Denny MW (2008) To bend a coralline: Effect of joint morphology on flexibility and stress amplification in an articulated calcified seaweed. J Exp Biol 211:3421-3432.
Martone PT (2007) Kelp versus Coralline: Cellular basis for mechanical strength in the wave-swept seaweed calliarthron (Corallinaceae, Rhodophyta). J Phycol 43(5):882-891.
Endean R (1961) The test of the ascidian, Phallusia mammillata. Q J Microsc Sci 102(1):107-117.
Fedonkin MA, Vickers-Rich P, Swalla BJ, Trusler P, Hall M (2012) A new metazoan from the Vendian of the White Sea, Russia, with possible affinities to the ascidians. Paleontol J 46(1):1-11.

Literature
& Links
Additional Imagery
 |
|



 ,
,